A metal cube has an edge length of 11.4 mm and a mass of 6.67 g
A metal cube has an edge length of 11.4 mm and a mass of 6.67 g. Calculate the density of the metal and refer to Table 1.4 to determine the likely identity of the metal.
Considering that density for a solid is generally g/cm3 , the edge length must be converted from mm to cm. This allows the formula for density to be used (D=m/v). The density of the metal cube is 4.51 g/cm3. This density would allow us to likely identify the metal cube as titanium.
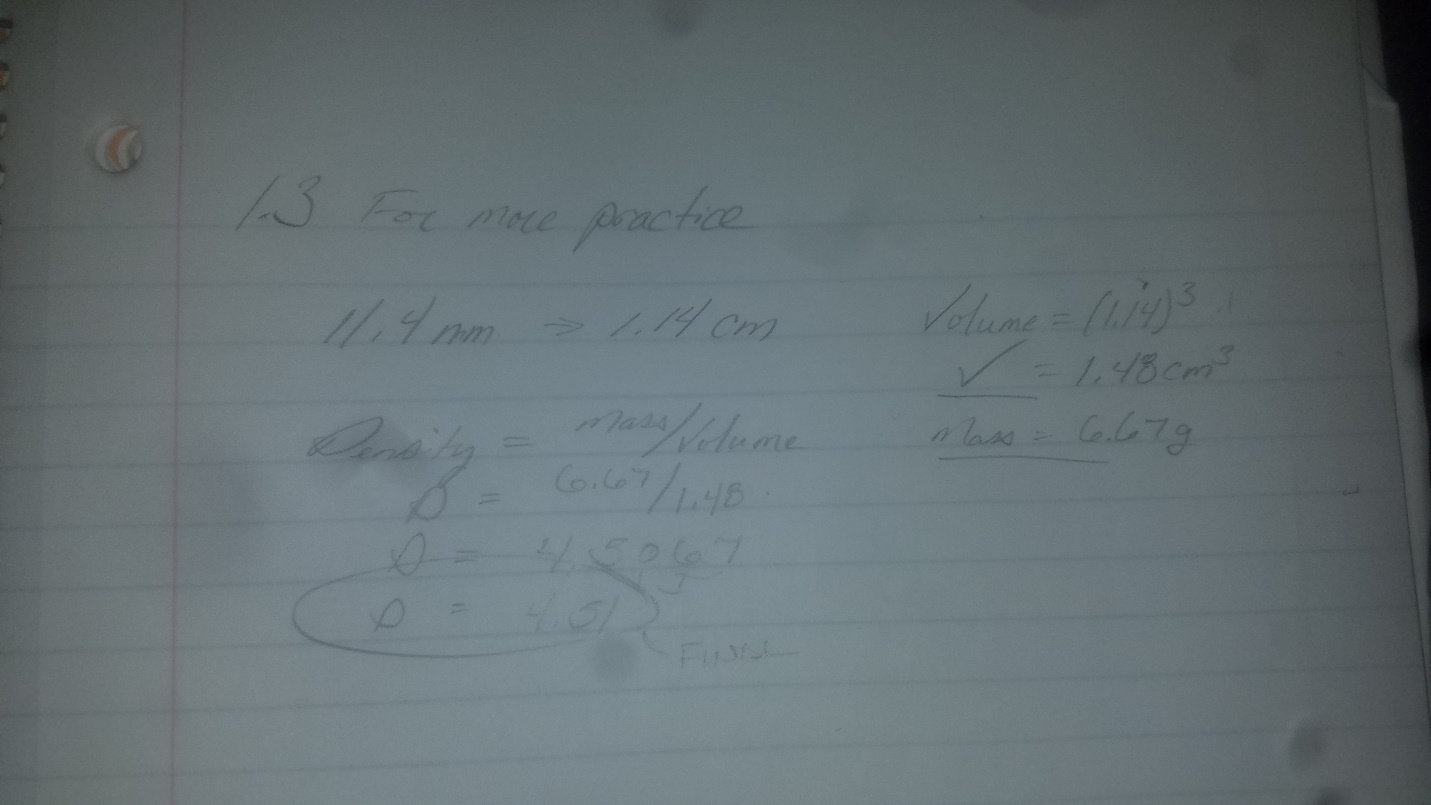
–Chapter 1. Cumulative Problem 81




